Inflammatory bowel disease (IBD) refers tochronic inflammatory disease of the colon or gastrointestinal tract, including ulcerative colitis (UC) and Crohn's disease (CD) Clinical symptoms of IBD include recurrent diarrhea, abdominal pain, intestinal bleeding/hematochezia, fever, and weight loss. The pathogenesis of IBD is not clear, and studies have shown that various factors such as genes, immune system, external environment and intestinal microorganisms are associated with the occurrence of disease. A variety of preclinical mouse models are currently used to study IBD, and disease models derived based on different induction methods have specific uses. The mouse enteritis model induced by dextran sulfate sodium (DSS) is the most widely used chemically-induced mouse IBD model. Acute ulcerative enteritis or chronic colitis isinduced by dissolving DSS in drinking water, which destroys intestinal epithelial cells in mice, releases cytokines by non-specific immune cells, and finally results in disruption of the integrity of the mucosal barrier. Animals experience significant weight loss, loose stools, hematochezia, and granulocyte infiltration, symptoms that are extremely similar to human ulcerative colitis in clinical symptoms and pathological features.
A stable DSS-induced IBD disease model protocol was established inC57BL/6 mice by Biocytogen, which can be used for preclinical studies and pharmacodynamic evaluation of inflammatory enteritis.
Experimental Animals:C57BL/6, 7-8 weeks old, female
Modeling reagent:DSS
Modeling method:dosing with DSS in drinking water for 7 consecutive days

Clinical assessment(DAI score)
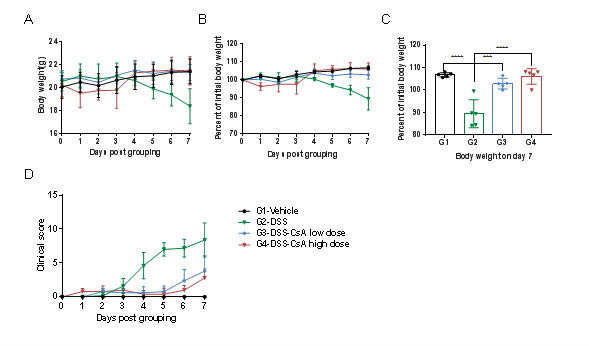
C57BL/6 mice were provided drinking water containing DSS for 7 consecutive days, and body weight changes were recorded throughout and scored clinically.(Fig. A-C) The change in body weight of animals in each group. (Fig. D) DAI score (diasease activity index) of animals in each group. The body weight of animals in the model group (G2-DSS) was decreased compared with that in the non-model group (G1-Vehicle), and the DAI score indicates increased disease severity; the body weight of the animals in the model group (G3-DSS-CSA low dose and G4-DSS-CSA high dose) did not change significantly, and the DAI score was slightly increased. This indicated that the DSS-induced inflammatory bowel disease model in C57BL/6 mice was successfully established, and CsA (cyclosporin A) relieved the clinical symptoms of IBD in a dose-dependent manner.

Comparison of colons in C57BL/6 mice with DSS-induced inflammatory bowel disease. (Fig. A) Colon length comparison. ( B) Colon weight to body weight ratio in each group of animals. (Fig. C), the ratio of colon weight to colon length in each group of animals. The experimental data showed that the colon weight and length were significantly abnormal in the model group (G2-DSS) compared with the non-model group (G1-Vehicle); the colon weight and length were improved in the CsA-treated groups (G3-DSS-CSA low dose and G4-DSS-CSA high dose) compared with the untreated mice. This suggests that CsA alleviates colonic symptoms in a DSS-induced inflammatory bowel disease model in mice.
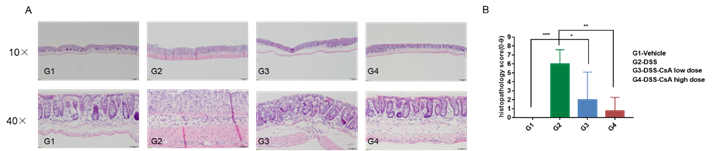
Photomicrograph of hematoxylin and eosin (H & E) stain cross section (10x and 40xmagnification).(A) H&E staining of the colon of animals in each group. (B) pathological scores of animals in each group. Compared with the control group (G1-Vehicle), the intestinal epithelial cells were destroyed in the G2 group, along with the integrity of the intestinal mucosal layer. In addition, inflammatory cell infiltration was observed in the modeled group (G2-DSS). In the CSA-treated IBD group, the colonic symptoms were significantly relieved in a dose-dependent manner.
Altogether, these data indicate that DSS colitis in C57BL/6 mice was successful, and cyclosporin A (CsA) could significantly alleviate DSS-induced inflammatory bowel disease colon lesions.
1. Elson, C.O., Sartor, R.B., Tennyson, G.S. & Riddell, R.H. Experimental models of inflammatory bowel disease. Gastroenterology 109, 1344-1367 (1995).
2. Kiesler, P., Fuss, I.J. & Strober, W. Experimental Models of Inflammatory Bowel Diseases. Cell Mol Gastroenterol Hepatol 1, 154-170 (2015).
3. Mourad, F.H., Yau, Y., Wasinger, V.C. & Leong, R.W. Proteomics in Inflammatory Bowel Disease: Approach Using Animal Models. Dig Dis Sci 62, 2266-2276 (2017).









