
C57BL/6N-Pdcd1tm3(PDCD1)Bcgen/Bcgen • 110019
Programmed death-1 (PD-1) is primarily expressed on T cells and primary B cells, and its ligands PD-L1 and PD-L2 are broadly expressed on antigen-presenting cells (APCs). PD-1 signaling negatively regulates immune responses, and PD-L1 is frequently detected in human tumors, where it can support immune evasion; therefore, blocking PD-1/PD-L1 interaction is a key strategy in cancer immunotherapy.
Validation data confirm the successful generation of PD-1 humanized mice (B-hPD-1 mice plus), demonstrating their suitability as a robust preclinical platform for pharmacodynamic, efficacy, and safety evaluation of anti-human PD-1 therapeutics.
Key Advantages
Validation
Applications
PD-1 humanized mice (B-hPD-1 mice plus) provide an in vivo platform for evaluating anti-human PD-1 therapeutics—including target engagement (e.g., nivolumab/pembrolizumab binding) and antitumor efficacy studies.
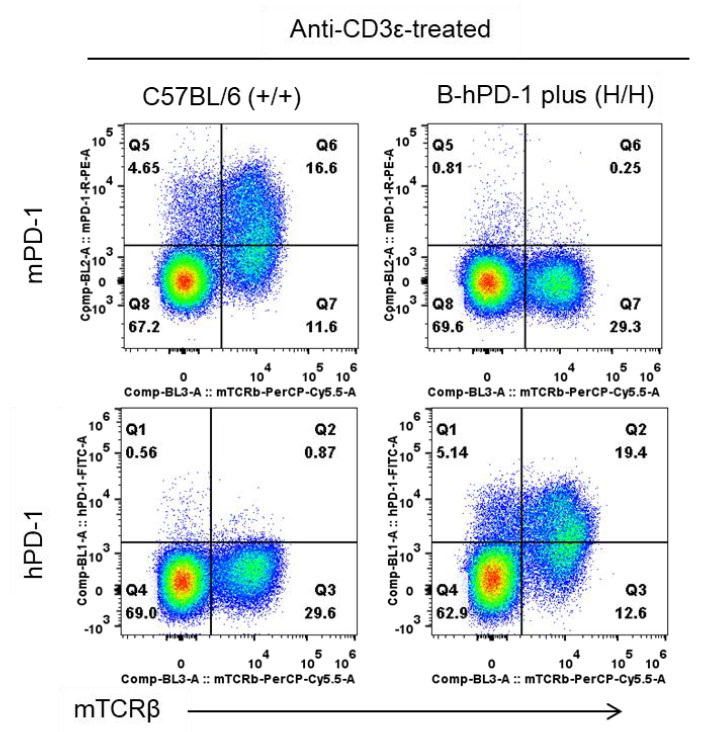
Species-specific PD-1 protein expression in PD-1 humanized mice (B-hPD-1 mice plus) was assessed by flow cytometry. Splenocytes were isolated from wild-type C57BL/6 mice (+/+) and homozygous PD-1 humanized mice (B-hPD-1 mice plus) (H/H) following in vivo anti-CD3ε stimulation, then stained with species-specific anti-PD-1 antibodies. Mouse PD-1 was detected only in wild-type C57BL/6 controls, whereas human PD-1 was exclusively detected in homozygous PD-1 humanized mice (B-hPD-1 mice plus) but not in wild-type mice.
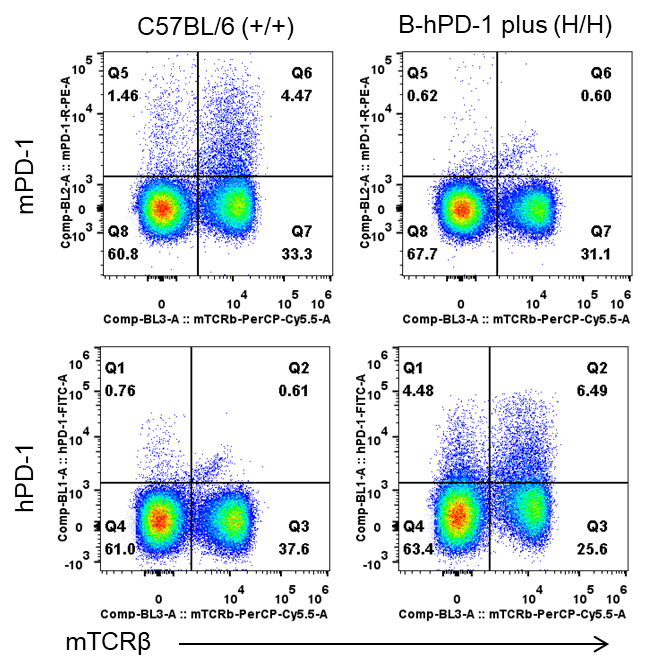
Species-specific PD-1 protein expression in PD-1 humanized mice (B-hPD-1 mice plus) was assessed by flow cytometry. Splenocytes were collected from wild-type C57BL/6 mice (+/+) and homozygous PD-1 humanized mice (B-hPD-1 mice plus, H/H), then analyzed using species-specific anti–PD-1 antibodies. Mouse PD-1 was detectable only in wild-type C57BL/6 controls, whereas human PD-1 was exclusively detectable in homozygous PD-1 humanized mice (B-hPD-1 mice plus) and absent in wild-type mice.
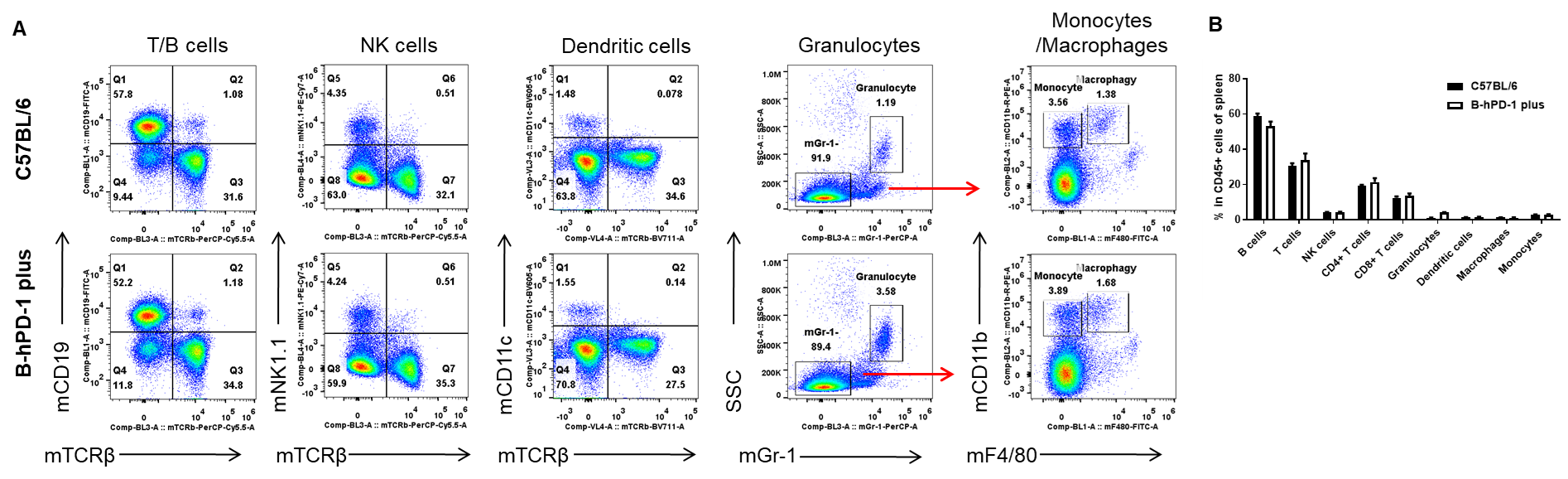
Species-specific analysis of spleen leukocyte subpopulations in PD-1 humanized mice (B-hPD-1 mice plus) by flow cytometry. Splenocytes were isolated from female wild-type C57BL/6 mice and PD-1 humanized mice (B-hPD-1 mice plus) (n=3, 7-week-old) and analyzed to profile major immune cell subsets. (A) Representative gating strategy used to identify T/B cells, NK cells, dendritic cells, granulocytes, and monocytes/macrophages from single live CD45⁺ cells, with lineage markers as indicated. (B) Quantification shows that the proportions of these leukocyte subsets in PD-1 humanized mice (B-hPD-1 mice plus) are comparable to those in C57BL/6 mice, indicating that PD-1 humanization does not measurably alter overall spleen leukocyte composition. Values are expressed as mean ± SEM.
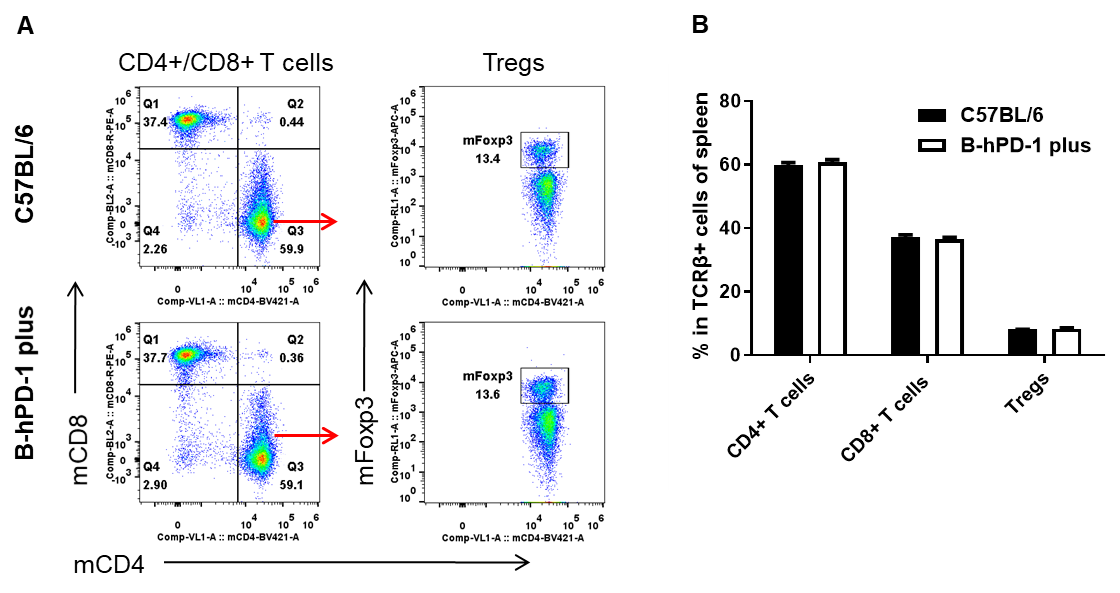
Species-specific analysis of spleen T-cell subsets in PD-1 humanized mice (B-hPD-1 mice plus) by flow cytometry. Splenocytes were collected from female C57BL/6 and PD-1 humanized mice (B-hPD-1 mice plus) (n=3, 7-week-old) and analyzed to profile T-cell subpopulations. (A) Representative FACS plots show gating of single live CD45⁺ cells followed by identification of TCRβ⁺ T cells and downstream CD4⁺, CD8⁺, and regulatory T cells (Tregs; Foxp3⁺). (B) Quantification indicates that the proportions of CD4⁺ T cells, CD8⁺ T cells, and Tregs in spleen are comparable between PD-1 humanized mice (B-hPD-1 mice plus) and C57BL/6 controls, suggesting that PD-1 humanization does not alter the overall distribution of major splenic T-cell subsets. Values are expressed as mean ± SEM.
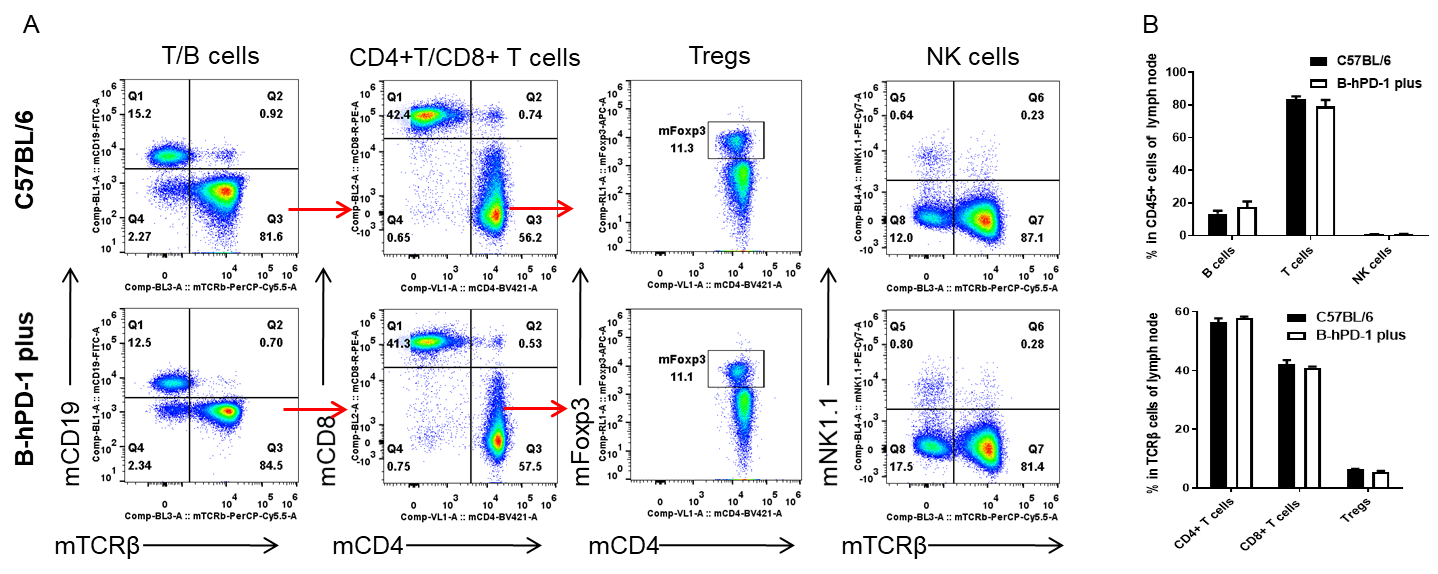
Species-specific analysis of lymph node leukocyte subsets in PD-1 humanized mice (B-hPD-1 mice plus) by flow cytometry. Lymph nodes were collected from female C57BL/6 and PD-1 humanized mice (B-hPD-1 mice plus, n=3, 7-week-old), and leukocytes were profiled by flow cytometry. (A) Representative gating plots show identification of major populations, including T/B cells, CD4⁺/CD8⁺ T cells, Tregs, and NK cells. Single live CD45⁺ cells were gated and analyzed using lineage markers as indicated. (B) Quantification shows that the proportions of B cells, T cells, CD4⁺ T cells, CD8⁺ T cells, Tregs, and NK cells in PD-1 humanized mice (B-hPD-1 mice plus) are comparable to those in C57BL/6 mice, indicating that PD-1 humanization does not alter lymph node immune cell composition. Values are expressed as mean ± SEM.
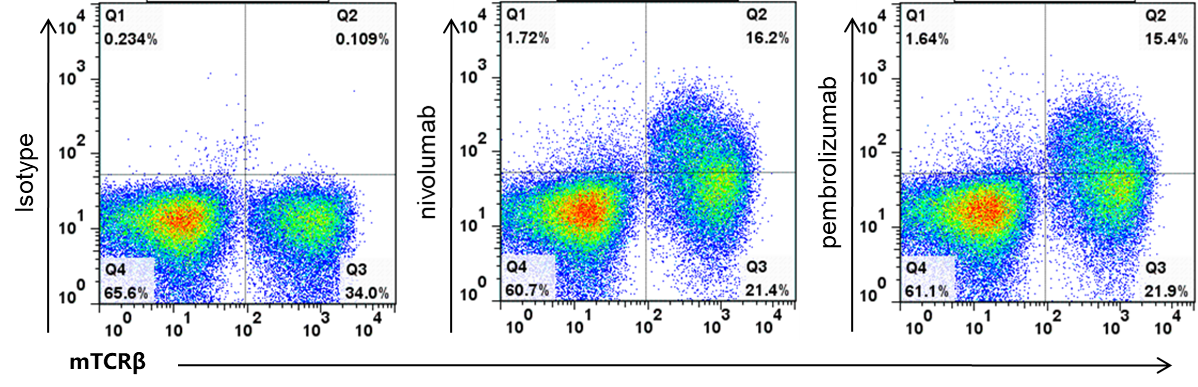
Binding validation of anti-human PD-1 antibodies in PD-1 humanized mice (B-hPD-1 mice plus) by flow cytometry. Splenocytes were isolated from female PD-1 humanized mice (B-hPD-1 mice plus) and analyzed by flow cytometry to assess human PD-1 accessibility on T cells. Single live CD45⁺ cells were gated, followed by identification of mTCRβ⁺ T cells as indicated. Compared with the isotype control, nivolumab and pembrolizumab showed specific binding to T cells from PD-1 humanized mice (B-hPD-1 mice plus), confirming that human PD-1 is functionally accessible for therapeutic antibody engagement.
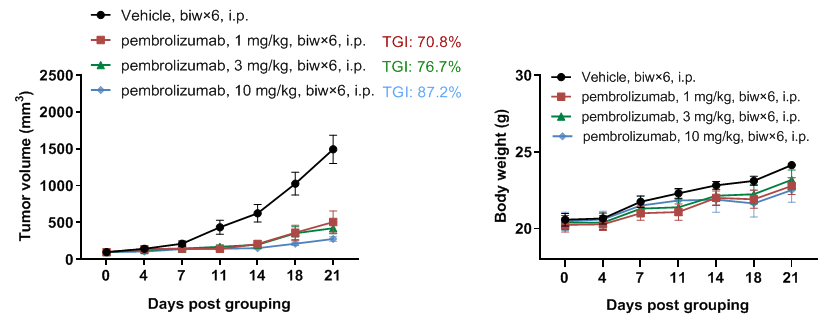
In vivo antitumor activity of anti-human PD-1 antibody pembrolizumab in PD-1 humanized mice (B-hPD-1 mice plus). (A) Murine colon cancer MC38 cells (5×105) were implanted subcutaneously into homozygous PD-1 humanized mice (B-hPD-1 mice plus; female, 7-week-old, n=5). Mice were grouped when tumor volume reached approximately 150 mm³, then treated intraperitoneally (i.p.) biweekly for 6 doses (biw×6) with vehicle or pembrolizumab (in house) at 1, 3, or 10 mg/kg according to the dosing schedule shown. Tumor volume was monitored throughout the study; pembrolizumab inhibited MC38 tumor growth with TGIs of 70.8% (1 mg/kg), 76.7% (3 mg/kg), and 87.2% (10 mg/kg). (B) Body weight was monitored during treatment to assess tolerability. As shown in panel A, pembrolizumab was efficacious in controlling tumor growth in PD-1 humanized mice (B-hPD-1 mice plus), demonstrating that PD-1 humanized mice (B-hPD-1 mice plus) provide a powerful preclinical model for in vivo evaluation of anti-human PD-1 antibodies. Values are expressed as mean ± SEM.
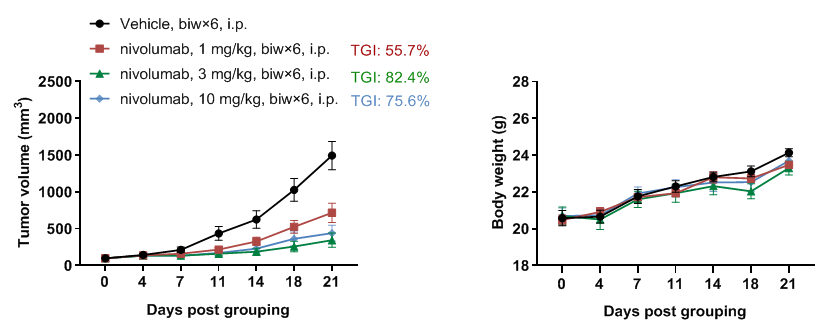
In vivo antitumor activity of anti-human PD-1 antibody nivolumab in PD-1 humanized mice (B-hPD-1 mice plus). (A) Nivolumab inhibited MC38 tumor growth in PD-1 humanized mice (B-hPD-1 mice plus). Murine colon cancer MC38 cells (5 × 10⁵) were subcutaneously implanted into homozygous PD-1 humanized mice (B-hPD-1 mice plus) (female, 7-week-old, n = 5). Mice were grouped when tumor volume reached approximately 150 mm³, then treated with nivolumab at the doses and schedules indicated in the panel. (B) Body weight changes were monitored throughout treatment. As shown in panel A, nivolumab was efficacious in controlling tumor growth in PD-1 humanized mice (B-hPD-1 mice plus), demonstrating that PD-1 humanized mice (B-hPD-1 mice plus) provide a powerful preclinical model for in vivo evaluation of anti-human PD-1 antibodies. Values are expressed as mean ± SEM.
Q1. How is species-specific PD-1 expression validated in PD-1 humanized mice (B-hPD-1 mice plus)?
Validation data demonstrate that mouse PD-1 is detectable only in wild-type C57BL/6 mice, whereas human PD-1 is exclusively expressed in homozygous PD-1 humanized mice (B-hPD-1 mice plus). Flow cytometry confirms human PD-1 expression on T cells, including after in vivo anti-CD3ε stimulation, verifying correct species-specific replacement.
Q2. Can clinically relevant anti-human PD-1 antibodies bind to PD-1 humanized mice (B-hPD-1 mice plus)?
Yes. Human PD-1 expressed on T cells in PD-1 humanized mice (B-hPD-1 mice plus) is functionally accessible. Therapeutic antibodies such as nivolumab and pembrolizumab specifically bind human PD-1 in this model, demonstrating its suitability for evaluating anti-human PD-1 antibody pharmacodynamics and efficacy.
Q3. Does humanization of PD-1 alter immune cell development in PD-1 humanized mice (B-hPD-1 mice plus)?
Flow cytometry profiling indicates that major leukocyte populations in spleen and lymph nodes—including T cells, B cells, NK cells, dendritic cells, granulocytes, monocytes/macrophages—and T-cell subsets (CD4⁺, CD8⁺, and Tregs) are comparable between PD-1 humanized mice (B-hPD-1 mice plus) and wild-type controls, supporting preserved immune system integrity.
Q4. What are the primary applications of PD-1 humanized mice (B-hPD-1 mice plus)?
PD-1 humanized mice (B-hPD-1 mice plus) are designed for in vivo evaluation of anti-human PD-1 therapeutics. In syngeneic tumor models such as MC38, treatment with anti-human PD-1 antibodies significantly inhibits tumor growth, making this model ideal for efficacy, pharmacodynamic, and combination immunotherapy studies.