
C57BL/6-Tnfsf8tm2(TNFSF8)Bcgen/Bcgen • 113224
Gene targeting strategy for B-hCD30L mice. The exons 1-4 of mouse Cd30l gene that encode extracellular domain were replaced by human counterparts in B-hCD30L mice. The genomic region of mouse Cd30l gene that encodes transmembrane domain and cytoplasmic portion was retained. The promoter and 5’UTR region of the mouse gene were also retained. The 3’UTR region of the mouse gene are replaced by human counterparts. The CD30L expression was driven by endogenous mouse Cd30l promoter, while mouse Cd30l gene transcription and translation will be disrupted.
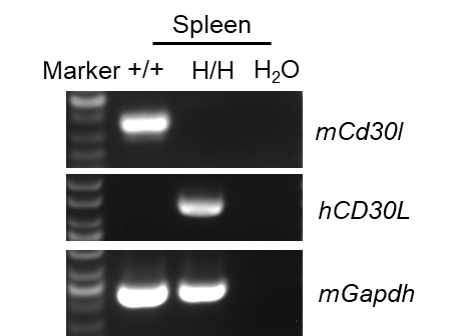
Species specific analysis of CD30L gene expression in wild-type C57BL/6JNifdc mice and homozygous humanized B-hCD30L mice by RT-PCR. Spleen was collected from wild-type C57BL/6JNifdc mice(+/+) and homozygous B-hCD30L mice(H/H). Mouse Cd30l mRNA was only detectable in wild-type C57BL/6JNifdc mice. Human CD30L mRNA was only detectable only in homozygous B-hCD30L mice, but not in wild-type C57BL/6JNidfc mice.
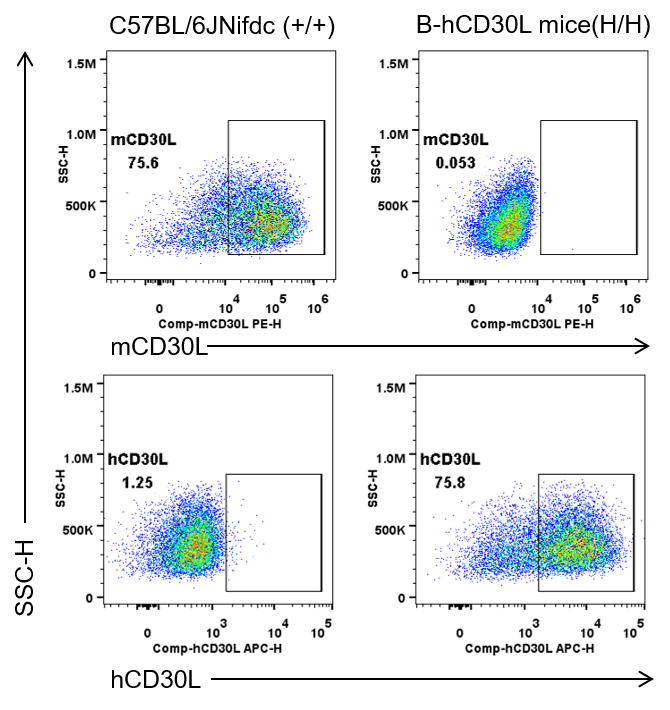
Strain specific CD30L expression analysis in wild-type C57BL/6JNifdc mice and homozygous humanized B-hCD30L mice by flow cytometry. Splenocytes were collected from wild-type C57BL/6JNifdc mice(+/+) and homozygous B-hCD30L mice(H/H). Splenocytes are incubated in a medium containing Cell Activation Cocktail (without Brefeldin A) (Biolegend, 423303) before analysis of CD30L surface expression on gated T cells. Protein expression was analyzed with anti-mouse CD30L antibody(Biolegend, 106405) and anti-human CD30L antibody(RD, FAB1028A) by flow cytometry. Mouse CD30L was exclusively detectable in wild-type C57BL/6JNifdc mice. Human CD30L was exclusively detectable in homozygous B-hCD30L mice, but not in wild-type C57BL/6JNifdc mice.
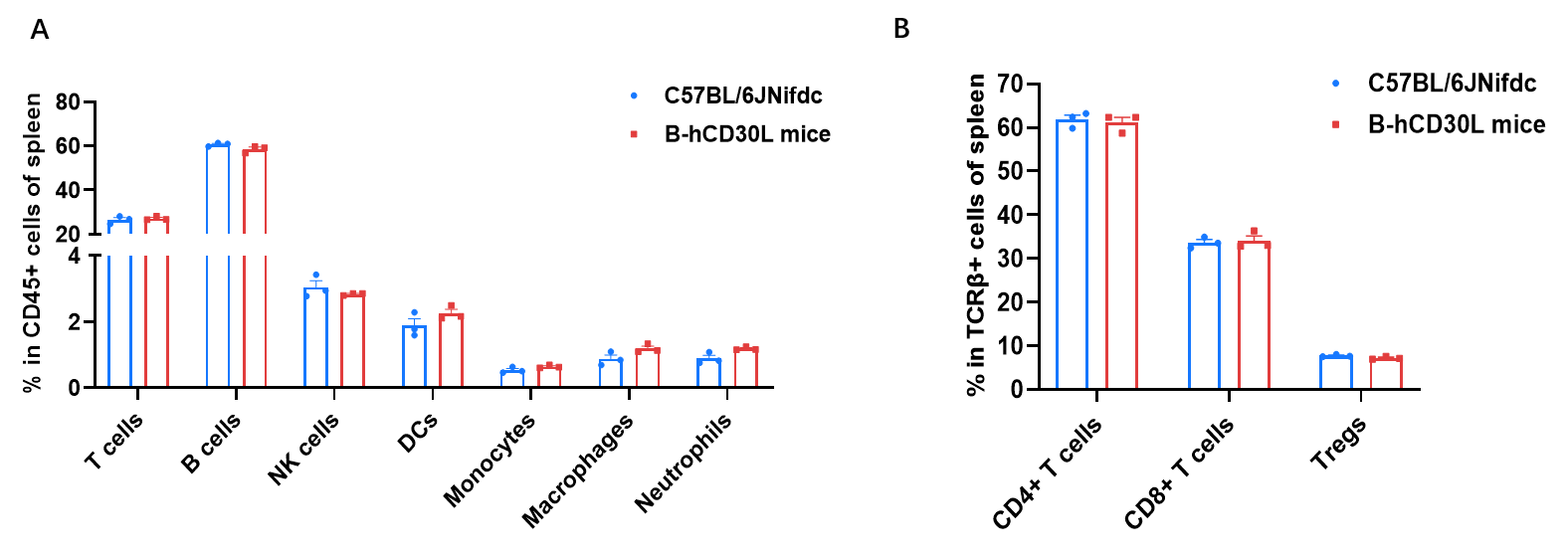
Frequency of leukocyte subpopulations in spleen by flow cytometry. Splenocytes were isolated from wild-type C57BL/6JNifdc mice and homozygous B-hCD30L mice (female, 6-week-old, n=3). A. Flow cytometry analysis of the splenocytes was performed to assess the frequency of leukocyte subpopulations. B. Frequency of T cell subpopulations. Frequencies of T cells, B cells, NK cells, DCs, neutrophils, monocytes, macrophages, CD4+ T cells, CD8+ T cells and Tregs in B-hCD30L mice were similar to those in C57BL/6JNifdc mice, demonstrating that humanization of CD30L does not change the frequency or distribution of these cell types in spleen. Values are expressed as mean ± SEM.
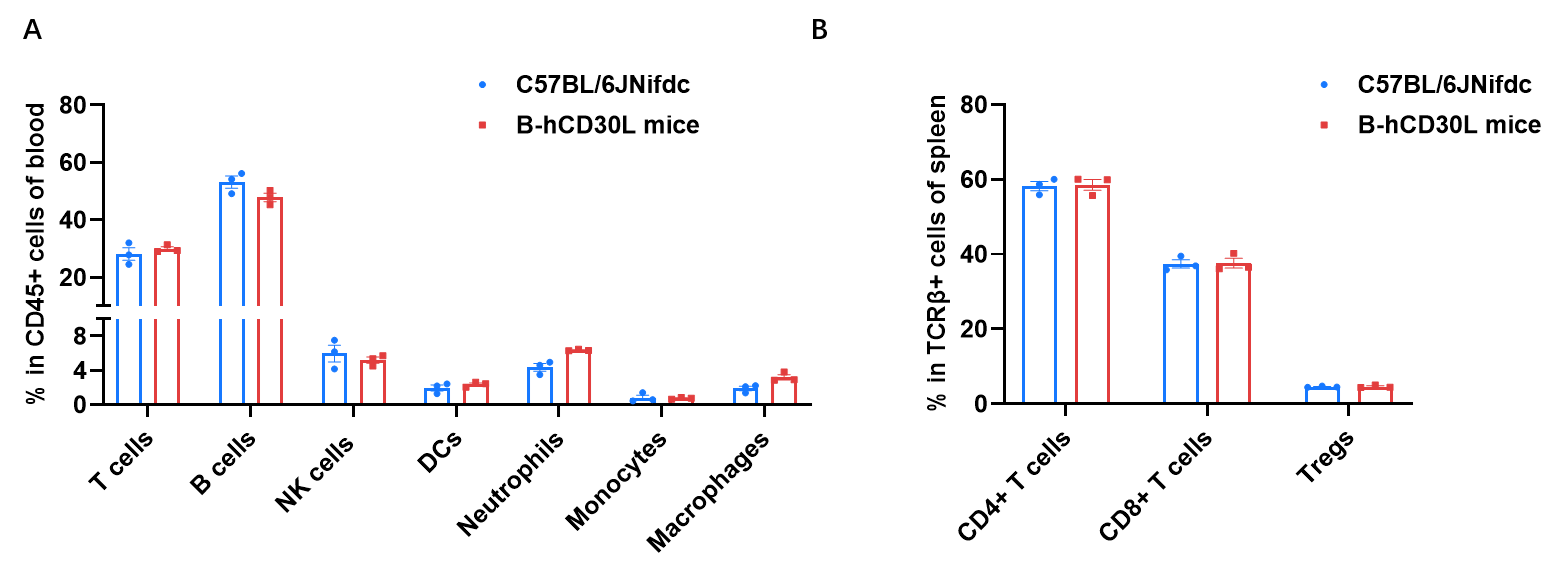
Frequency of leukocyte subpopulations in blood by flow cytometry. Blood cells were isolated from wild-type C57BL/6JNifdc mice (female, n=3, 6-week-old) and homozygous B-hCD30L mice (female, n=3, 6-week-old). A. Flow cytometry analysis of the blood cells was performed to assess the frequency of leukocyte subpopulations. B. Frequency of T cell subpopulations. Percentages of T cells, B cells, NK cells, dendritic cells, neutrophils, monocytes, macrophages, CD4+ T cells, CD8+ T cells and Tregs in B-hCD30L mice were similar to those in C57BL/6JNifdc mice, demonstrating that humanization of CD30L does not change the frequency or distribution of these cell types in blood. Values are expressed as mean ± SEM.
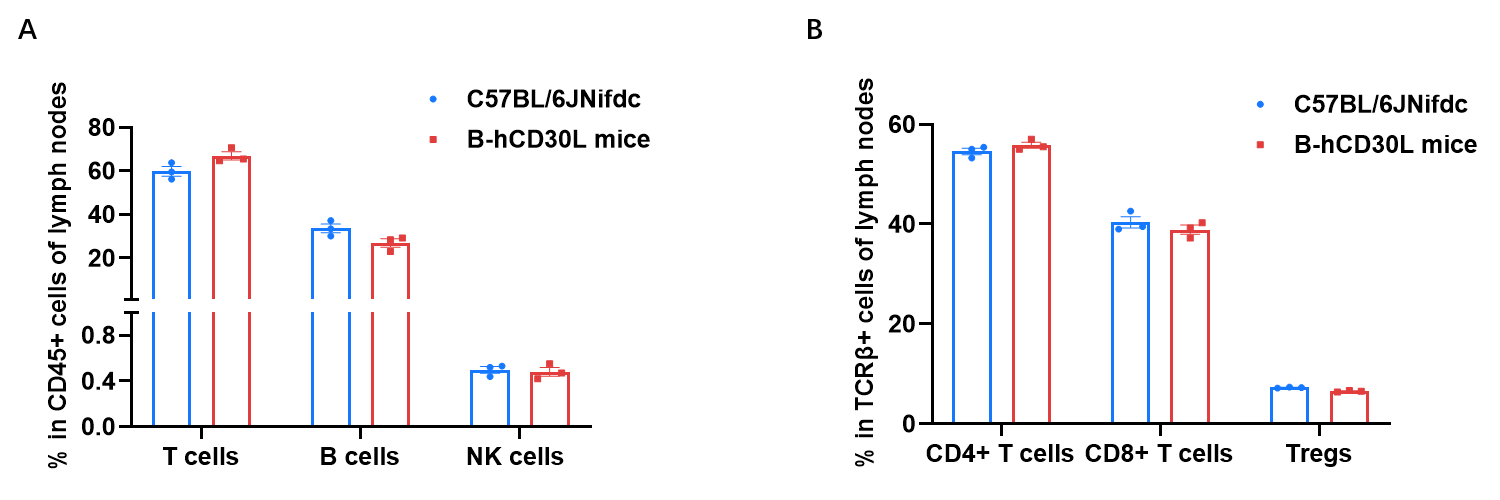
Frequency of leukocyte subpopulations in blood by flow cytometry. Lymph nodes cells were isolated from wild-type C57BL/6JNifdc mice (female, n=3, 6-week-old) and homozygous B-hCD30L mice (female, n=3, 6-week-old). A. Flow cytometry analysis of the lymph nodes cells was performed to assess the frequency of leukocyte subpopulations. B. Frequency of T cell subpopulations. Percentages of T cells, B cells, NK cells, CD4+ T cells, CD8+ T cells and Tregs in B-hCD30L mice were similar to those in C57BL/6JNifdc mice. Values are expressed as mean ± SEM.
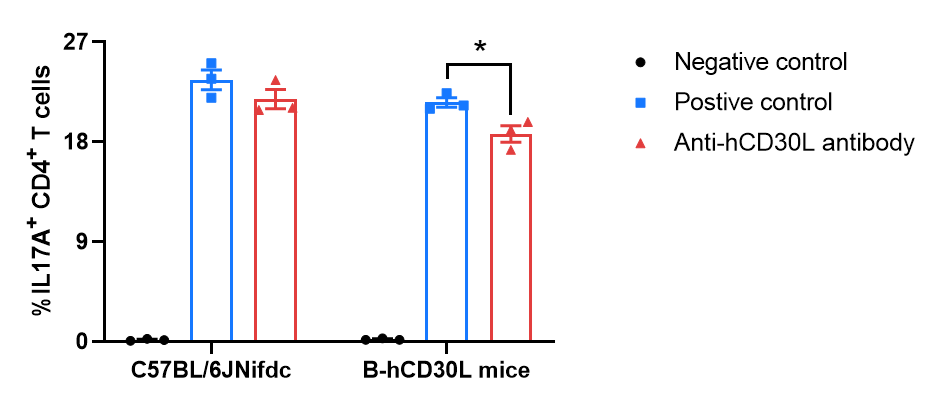
Strain specific mIL17A expression analysis in wide-type C57BL/6JNifdc and homozygous B-hCD30L mice by flow cytometry. Naïve CD4+ T cells were sorted from the splenocytes of wild-type C57BL/6JNifdc mice (+/+) and homozygous B-hCD30L mice (H/H), then the percentage of mIL17A positive cells were assessed after 72 h of incubation with rmIL6, rmIL23, rmTGF-β1, anti-mouse IFN-γ antibody and anti-mouse IL4 antibody in combination with bead-associated CD3 and CD28 mAbs. The anti-human CD30L antibody PRA052 (in house) could reduce the percentage of IL17A+ CD4+ T cells in B-hCD30L mice during Th17 cells differentiation, as PRA052 only recognizes human and does not recognize mice.

Mouse IL17A and IL23 expression analysis in wide-type C57BL/6JNifdc and homozygous B-hCD30L mice by ELISA. Naïve CD4+ T cells were sorted from the splenocytes of wild-type C57BL/6JNifdc mice (+/+) and homozygous B-hCD30L mice (H/H), and were assessed after 72 h of incubation with rmIL6, rmIL23, rmTGF-β1, anti-mouse IFN-γ antibody and anti-mouse IL4 antibody in combination with bead-associated CD3 and CD28 mAbs. The protein expression of mouse IL17A and mouse IL23 in cell culture supernatant were measured using the mouse IL17A ELISA kit (ProteinTech, KE10020) and mouse IL23 ELISA kit (R&D, M2300). The anti-human CD30L antibody PRA052 (in house) could reduce the production of mouse IL17A and mouse IL23 in B-hCD30L mice during Th17 cells differentiation, as PRA052 only recognizes human and does not recognize mice.

T-cell-dependent antibody response assay in homozygous B-hCD30L mice. B-hCD30L mice were used for the TDAR assay to evaluate the efficacy of the anti-hCD30L antibody (provided by the client). On Day 0, the mice were subcutaneously injected with 200 μg of KLH for immunization, another subcutaneous injection of 200 μg KLH was administered for boosting on Day 14. On Day 21, serum was collected from the mice to assess the levels of KLH-specific IgG. Treatment with the anti-hCD30L antibody (provided by the client) significantly reduced the level of KLH-specific IgG in mouse serum, indicating that the anti-hCD30L antibody could block T cell-dependent B cell activation. This data validated the effectiveness of B-hCD30L mice as a preclinical evaluation model for anti-hCD30L antibodies. Values are expressed as mean ± SEM. Significance was determined by unpaired t test. *P < 0.05.
Note: This experiment was conducted by the client using B-hCD30L mice.