
BALB/cCrSlcNifdc-Egfrtm2(EGFR)Bcgen/Bcgen • 113865
이 페이지에서
Gene targeting strategy for B-hEGFR mice(C). A chimeric CDS that encode human EGFR extracellular domain, mouse EGFR signal peptide, transmembrane and cytoplasmic domain, followed by mouse 3’UTR-STOP was inserted right after mouse Egfr exon2. The chimeric EGFR protein expression was driven by endogenous mouse Egfr promoter, while mouse Egfr gene transcription and translation will be disrupted.
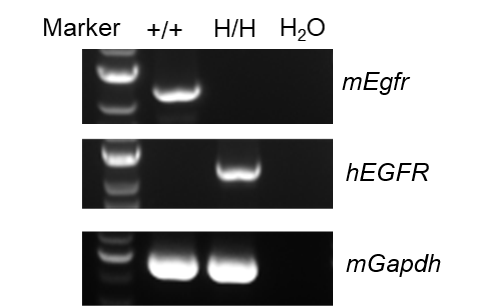
Strain specific analysis of EGFR mRNA expression in wild-type BALB/cCrSlcNifdc and B-hEGFR mice(C) by RT-PCR. Liver RNA were isolated from wild-type BALB/cCrSlcNifdc (+/+) and homozygous B-hEGFR mice(C) (H/H), then cDNA libraries were synthesized by reverse transcription, followed by PCR with mouse or human EGFR primers. mouse Egfr mRNA was only detectable in wild-type BALB/cCrSlcNifdc. Human EGFR mRNA was exclusively detectable in homozygous B-hEGFR mice(C) but not in wild-type BALB/cCrSlcNifdc.
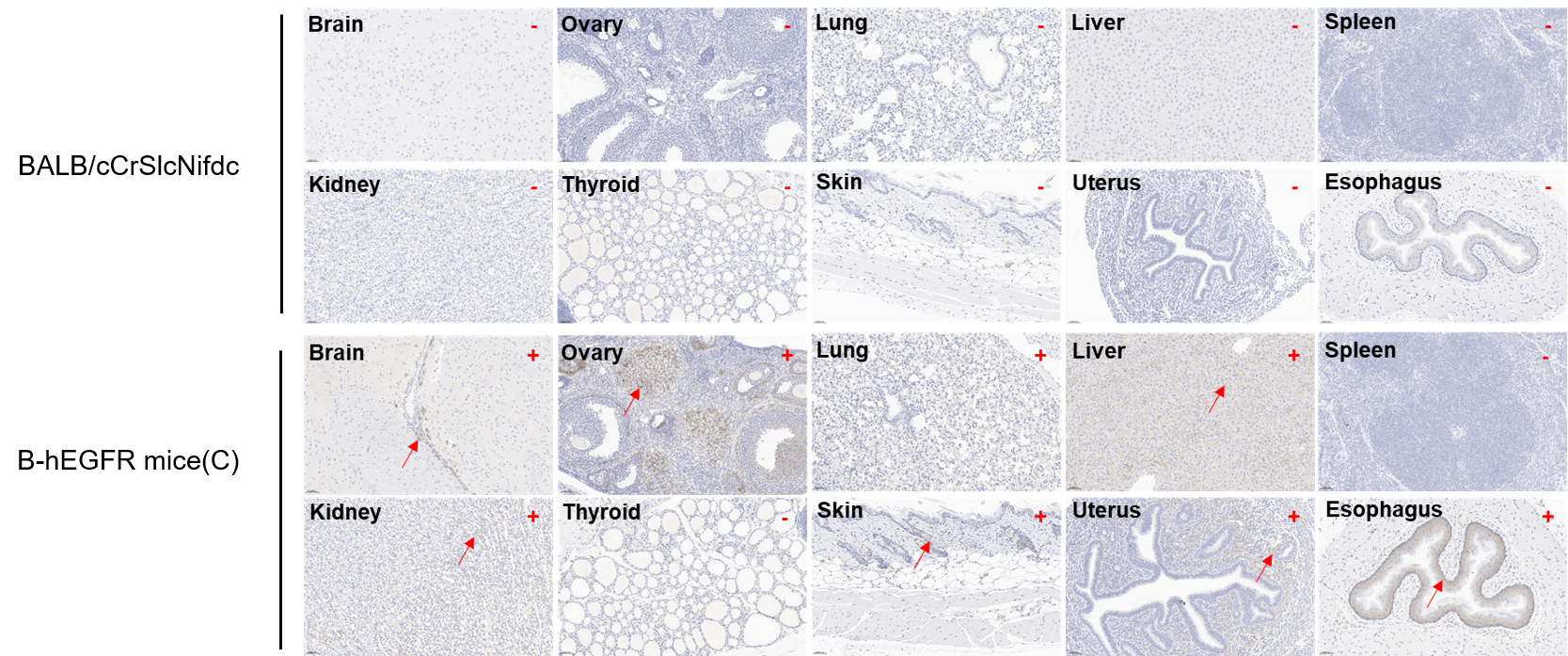
Immunohistochemical (IHC) analysis of EGFR expression in B-hEGFR mice(C). The ten tissues were collected from wild-type BALB/cCrSlcNifdc mice and B-hEGFR mice(C) (female, 6-week-old), analyzed by IHC with anti-EGFR (Invitrogen, MA5-49312). Human EGFR was detectable in brain, ovary, lung, liver, kidney, skin, uterus, and esophagus of B-hEGFR mice(C) but not in spleen and thyroid. Meantime, human EGFR was not detectable in tissues of BALB/cCrSlcNifdc mice. The arrow indicates tissue cells with positive EGFR staining (brown). “+” indicates that the tissue is positive, and “-” indicates that the tissue is negative.
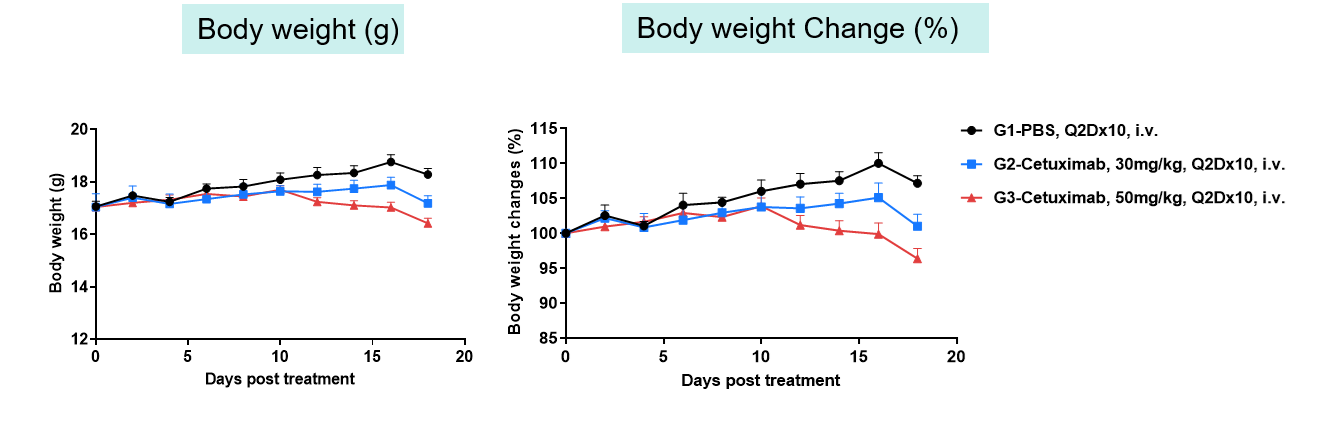
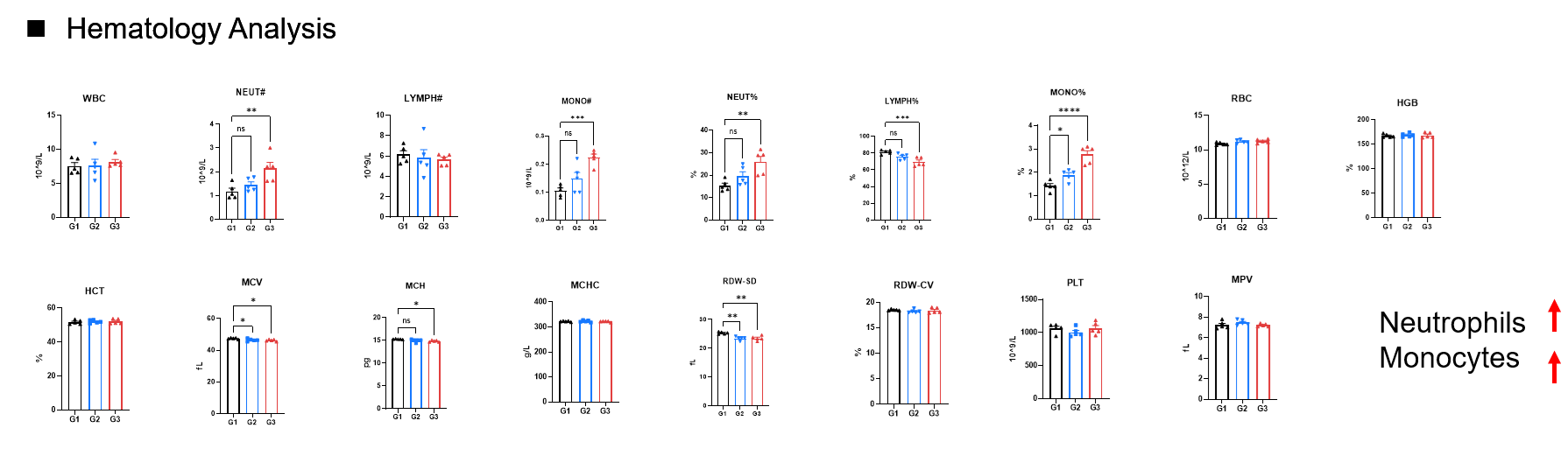
Toxicity analysis of anti-human EGFR antibody in B-hEGFR mice(C). Anti-human EGFR antibody cetuximab (Commercialized) were intravenously injected into B-hEGFR mice (female, 5 weeks old, n=5). Mice were weighed every two days, and their condition was observed daily. At the end of the experiment, blood samples were collected for complete blood count test. Additionally, tissue samples were collected from the oral mucosa, muzzle skin, abdominal skin, liver, esophagus, stomach, duodenum, jejunum, ileum, uterus, and ovary, and then subjected to pathological analysis. Values are expressed as mean ± SEM.
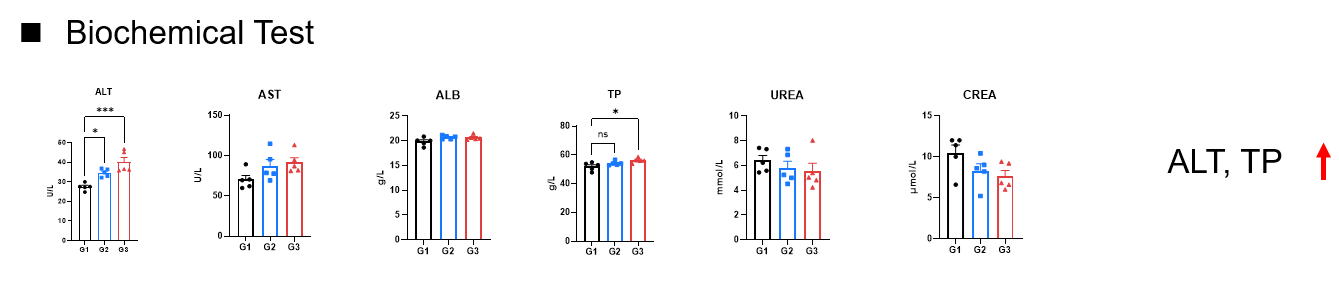
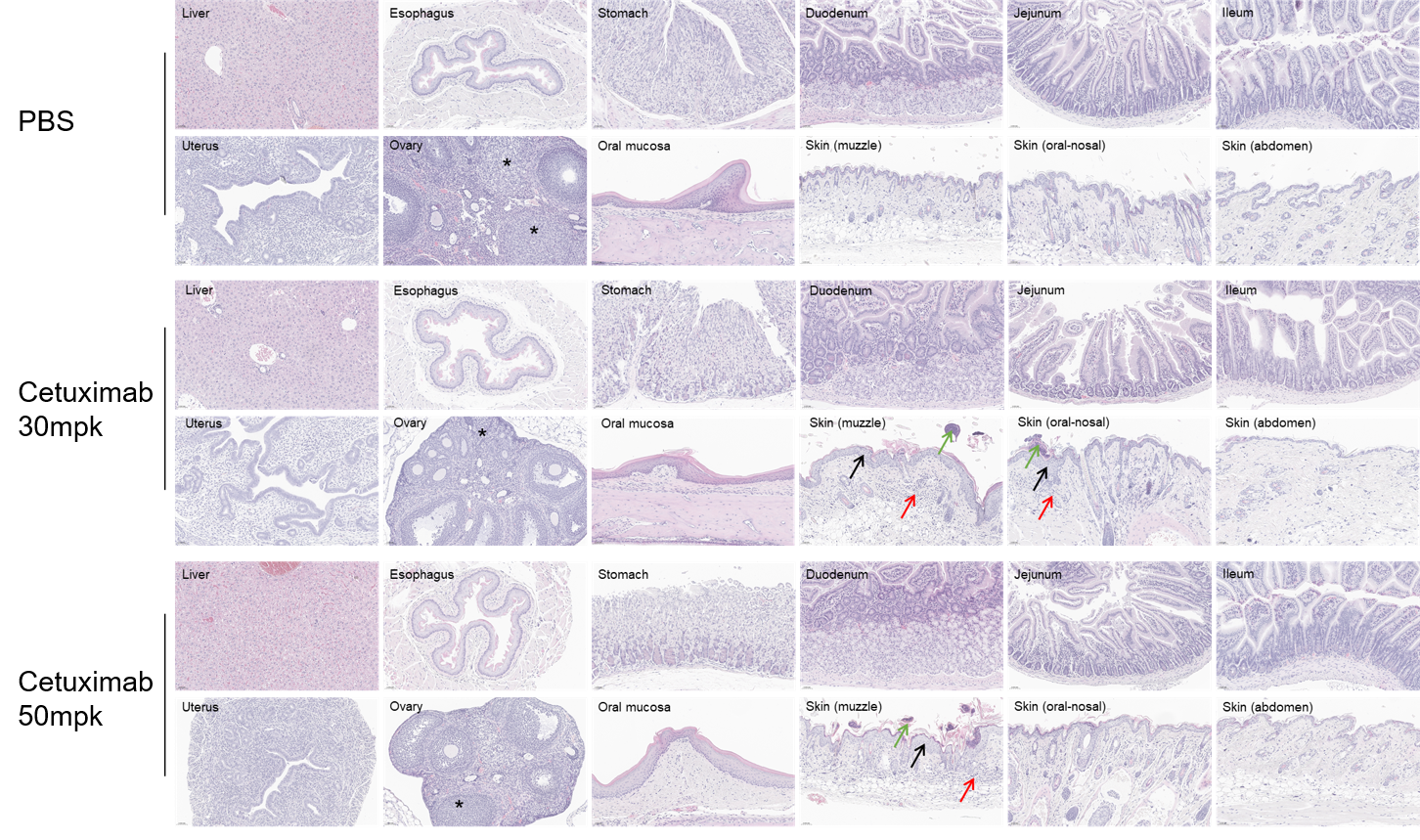
Pathological alterations in cetuximab-treated mice compared with PBS control. Cetuximab-treated mice showed epidermal hyperplasia with scab formation, and mixed inflammatory cell infiltration in the skin (muzzle), occasional in the skin (oral-nosal). Ovaries displayed a reduced number of corpora lutea. The incidence of pathological changes in the skin (around the eye) and ovaries were increased in the high dose group compared to those in the low dose group. These changes are considered related to cetuximab treatment.