
C57BL/6JNifdc-Scn9atm1(SCN9A)Bcgen/Bcgen • 111373
Gene targeting strategy for B-hSCN9A mice. The exons 2-27 of mouse Scn9a gene that encode the whole molecule (ATG to STOP codon) were replaced by human counterparts in B-hSCN9A mice. The promoter, 5’UTR and 3’UTR region of the mouse gene are retained. The human SCN9A expression is driven by endogenous mouse Scn9a promoter, while mouse Scn9a gene transcription and translation will be disrupted.
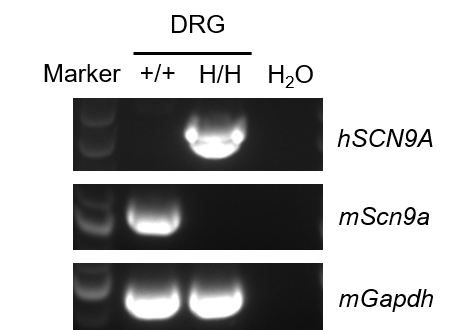
Strain specific analysis of SCN9A mRNA expression in wild-type C57BL/6JNifdc mice and homozygous B-hSCN9A mice by RT-PCR. Dorsal root ganglia (DRG) RNA was isolated from wild-type C57BL/6JNifdc mice (+/+) and homozygous B-hSCN9A mice (H/H), then cDNA libraries were synthesized by reverse transcription, followed by PCR with mouse or human SCN9A primers. Human SCN9A mRNA was detectable only in homozygous B-hSCN9A mice but not in wild-type mice. Mouse Scn9a mRNA was detectable only in wild-type C57BL/6JNifdc mice.
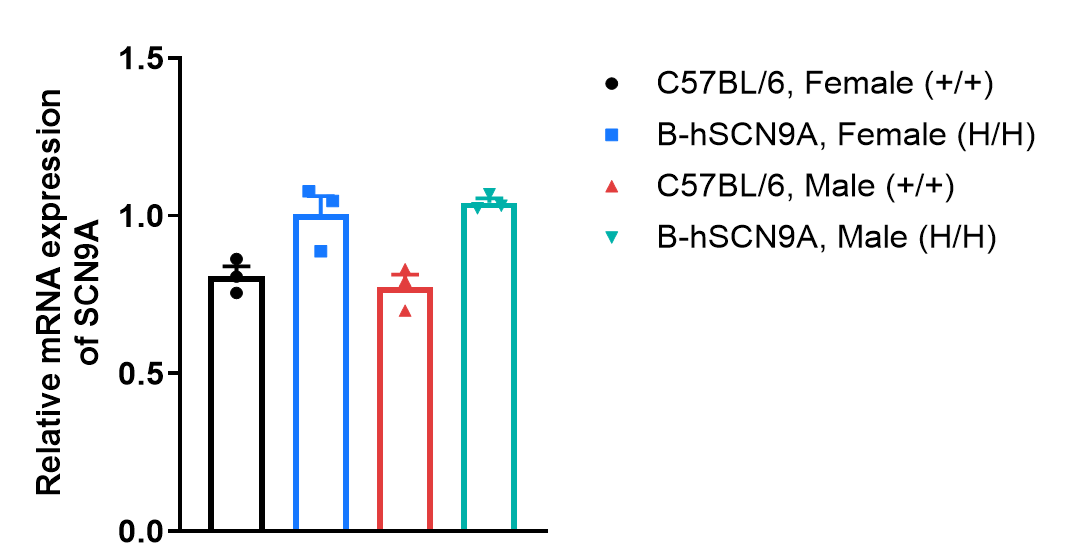
Species specific analysis of SCN9A gene expression in wild-type C57BL/6 mice and homozygous B-hSCN9A mice by RT-qPCR. Dorsal Root Ganglion (DRG) was collected from male and female wild-type C57BL/6 mice (+/+) and homozygous B-hSCN9A mice (H/H) (6-week-old, n=3). The mRNA expression of human SCN9A in homozygous B-hSCN9A mice was similar to that in wild-type C57BL/6 mice, demonstrating that introduction of human SCN9A in place of its mouse counterpart does not change the SCN9A expression. Values are expressed as mean ± SEM.
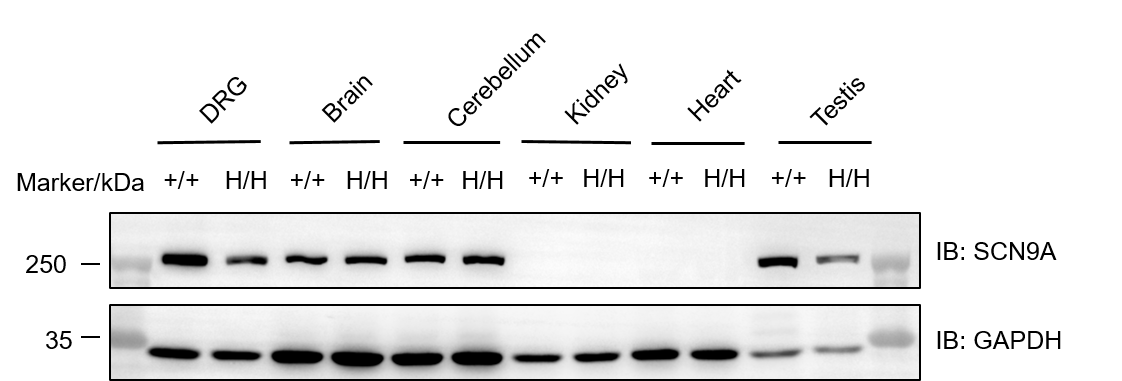
Western blot analysis of SCN9A protein expression in wild-type C57BL/6JNifdc mice and homozygous B-hSCN9A mice. Various tissue lysates were collected from wild-type C57BL/6JNifdc mice (+/+) and homozygous B-hSCN9A mice (H/H), and then analyzed by western blot with anti-Nav1.7 (SCN9A) Antibody. 40 μg total proteins were loaded for western blotting analysis. SCN9A protein was detected in DRG, brain, cerebellum and testis of both homozygous B-hSCN9A mice and wild-type mice.